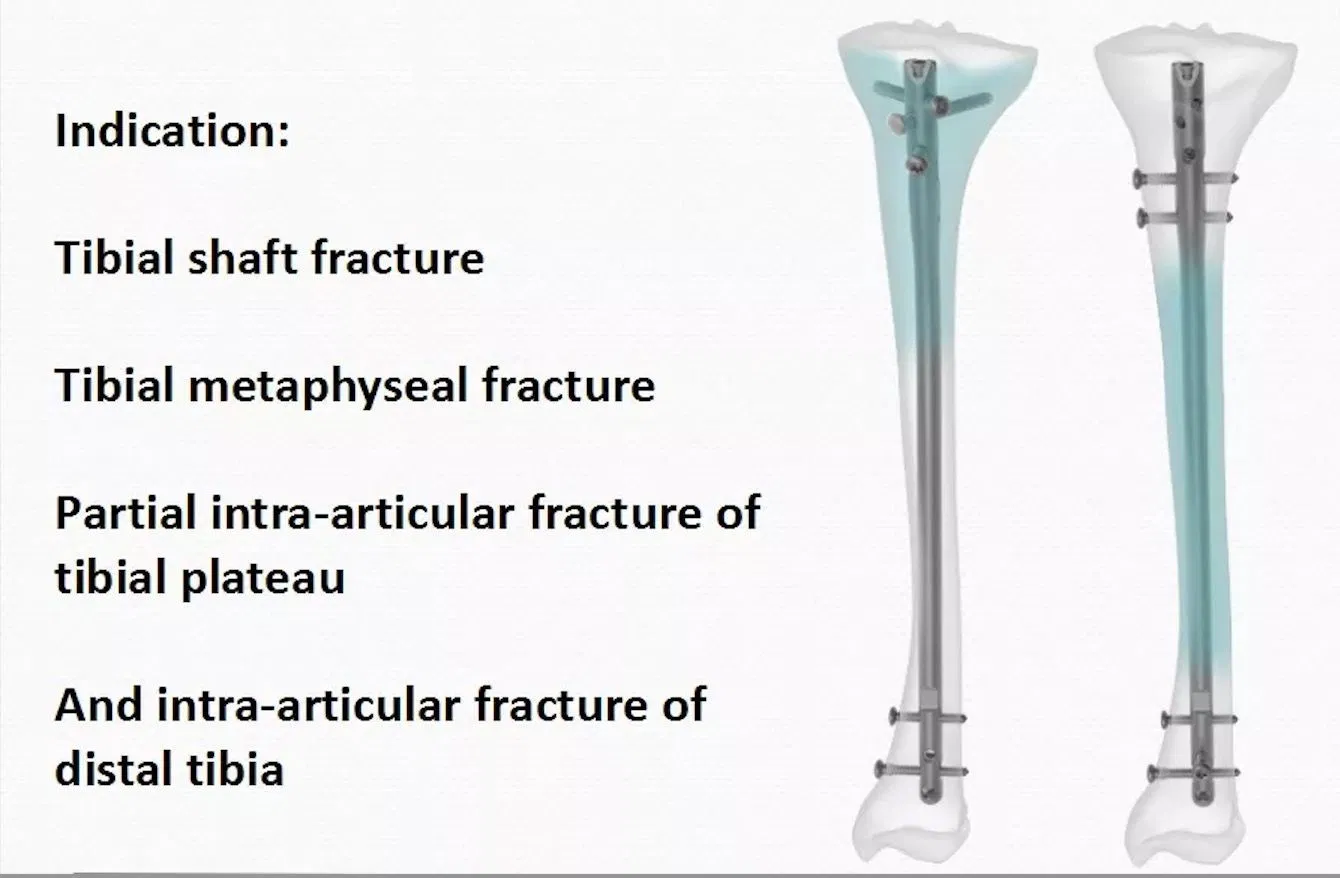




1 / 5
 Medisage Medical
Medisage Medical










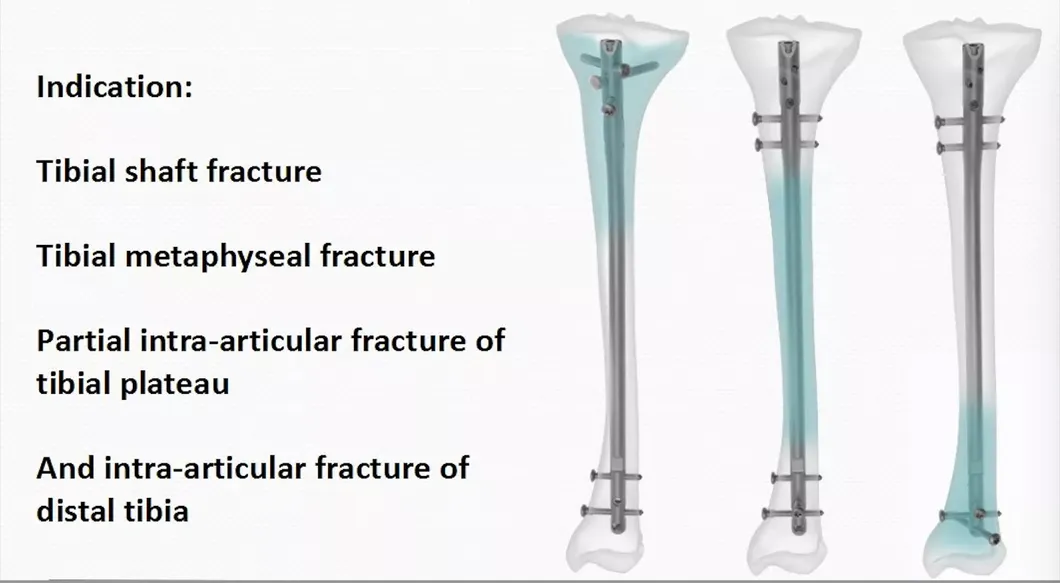

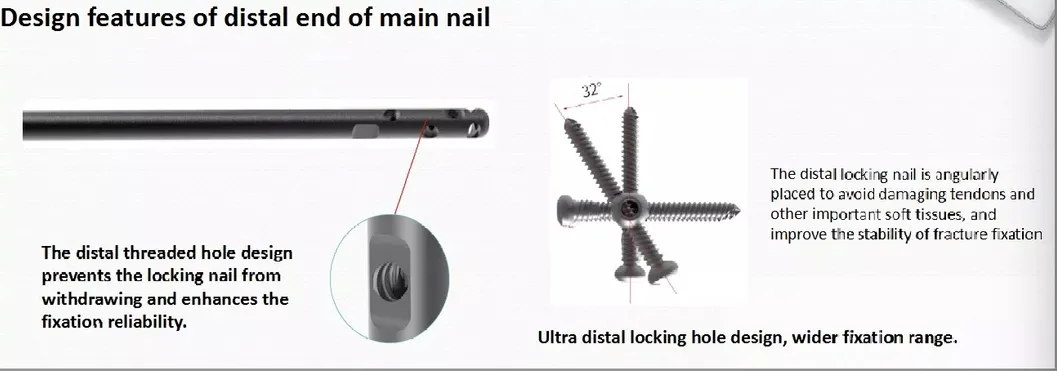
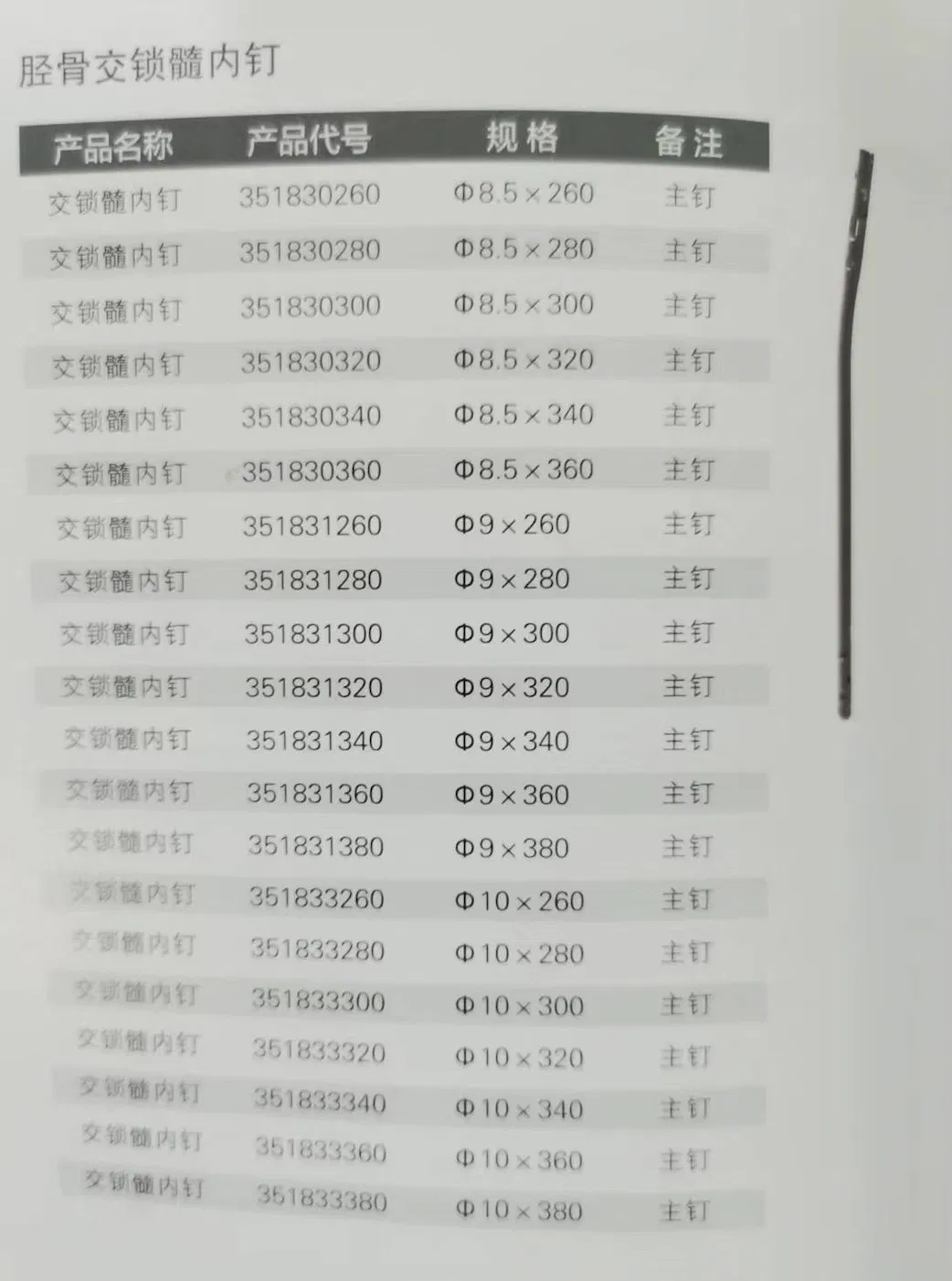


*All the prices are only for reference. Exact price will be based on the latest quotation.



