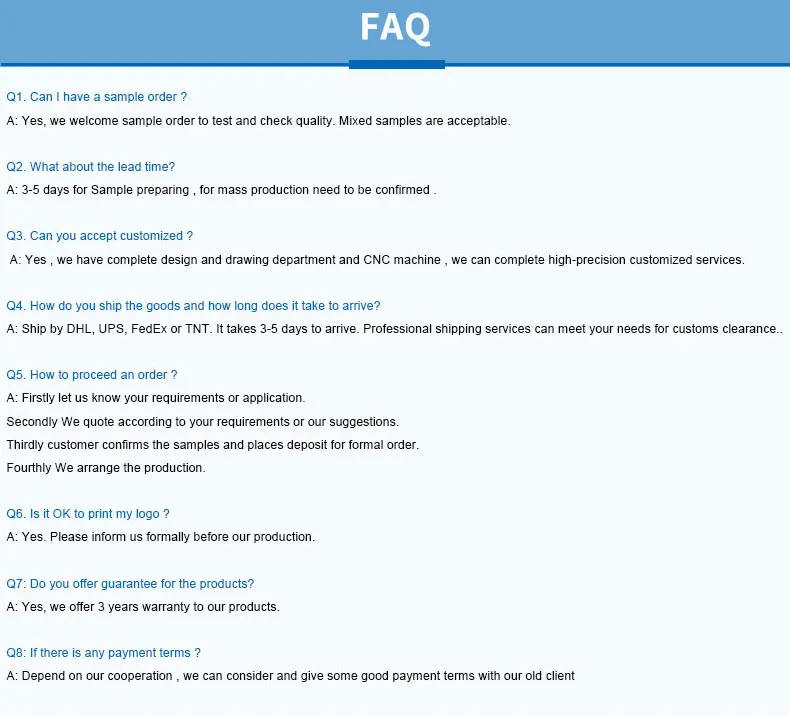
1 / 5
 Medisage Medical
Medisage Medical











The 5.5/6.0 screw-rod system is designed for spinal internal fixation. Featuring double/single thread options, this mature implant system offers flexibility with both sterile and non-sterile packaging choices.
This system is essential for addressing various spinal conditions that cause instability:
| Product name | Spinal Internal Fixation 5.5/6.0 Double Thread Mono Pedicle Spinal Screw |
| Material | Titanium |
| Color | Gold, Blue, Grey, Pink, etc |
| Related Rod | 5.5/6.0mm |
| Diameter | 4.5/5.0/5.5/6.0/6.5/7.0 mm |
| Length | 30/35/40/45/50/55/60 mm |
| Certificates | CE/ISO 13485 |
| Package | Non Sterilized / Sterilized |





Our Equipment: