

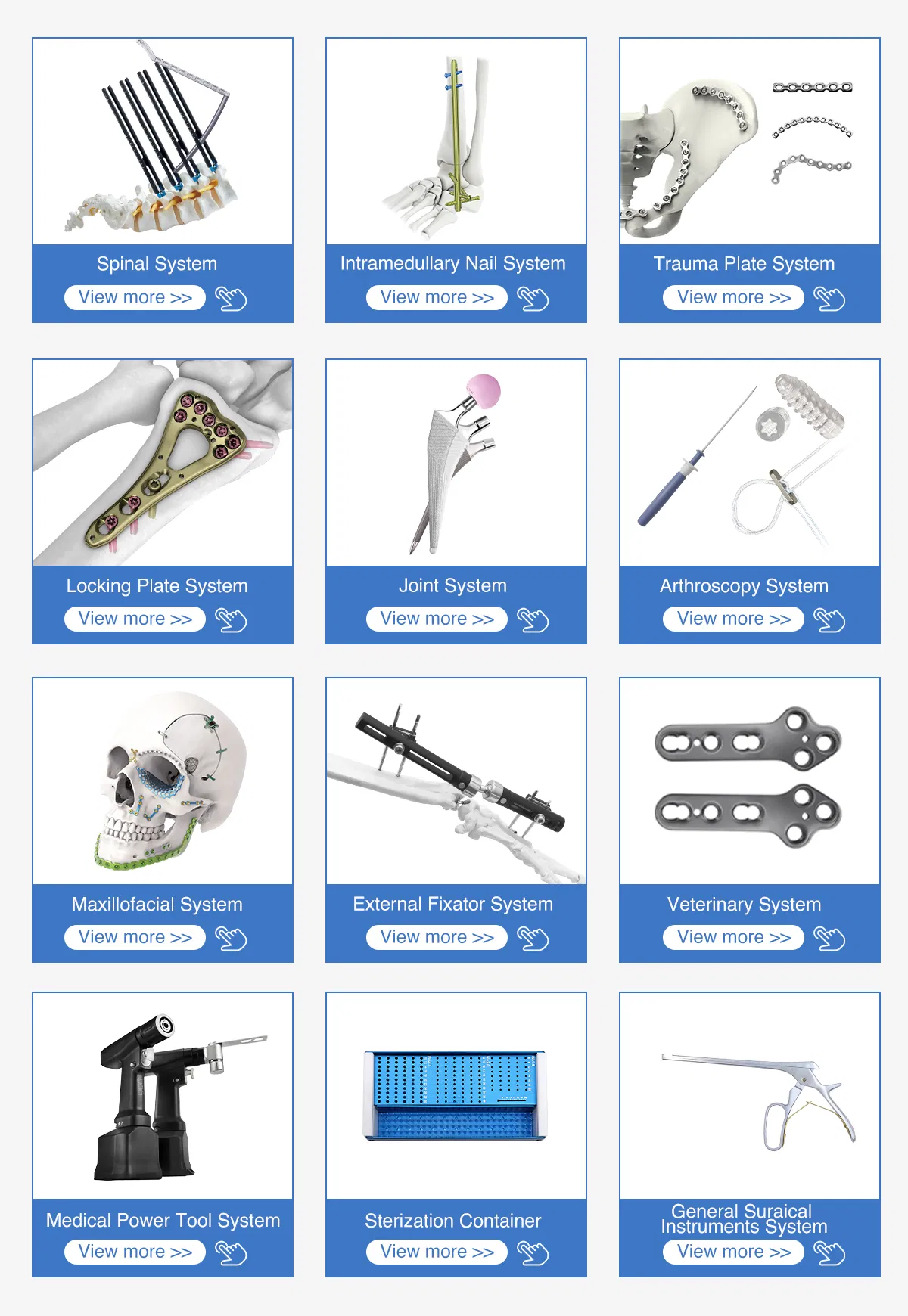
 Medisage Medical
Medisage Medical










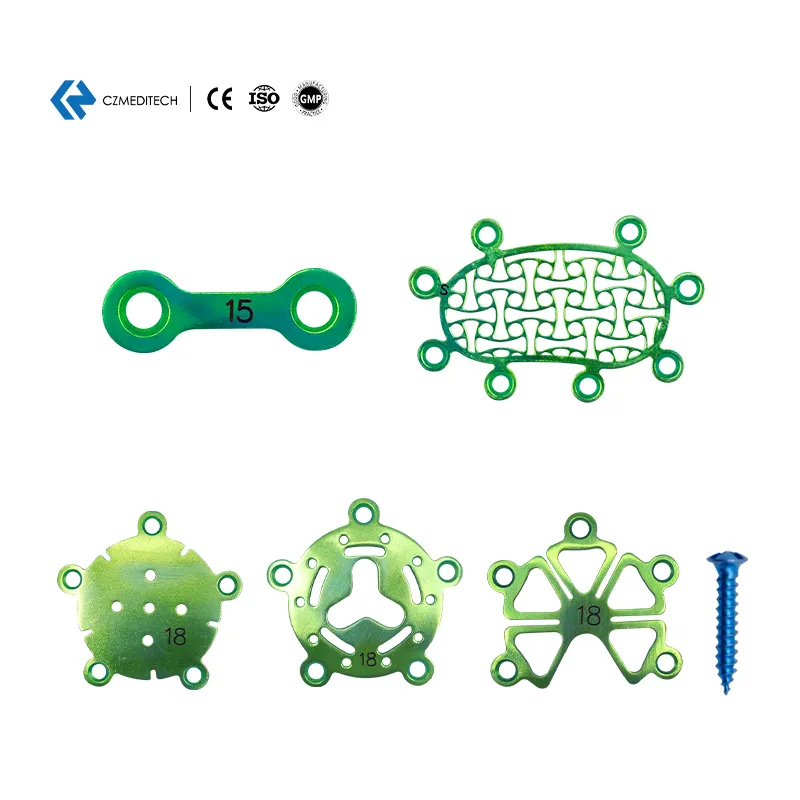
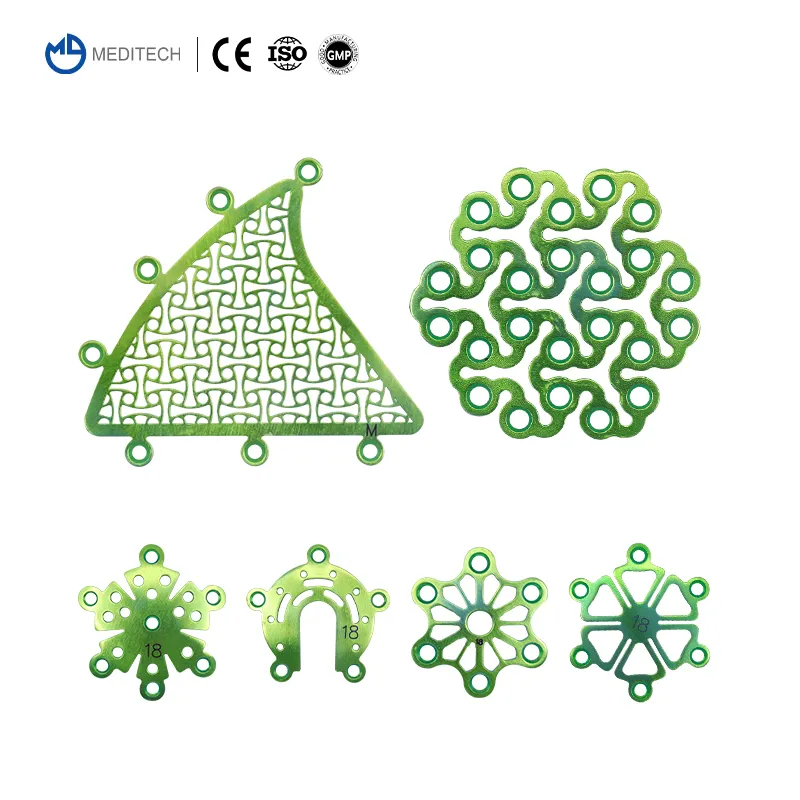
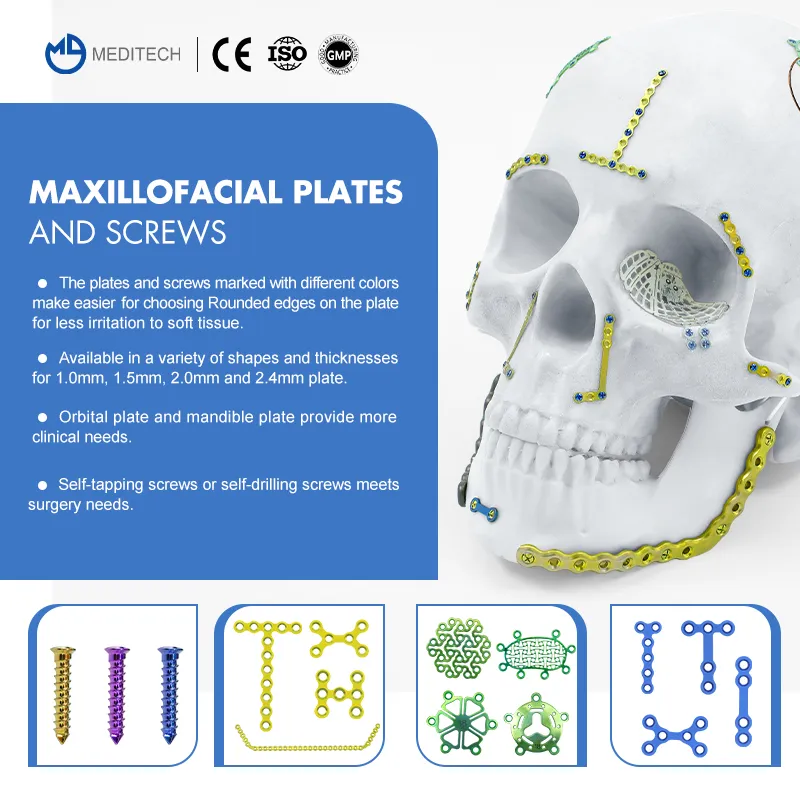
0.6mm Neurosurgery Restoration and Reconstruction System | CE Certified Titanium Cranial Fixation Implant Kit
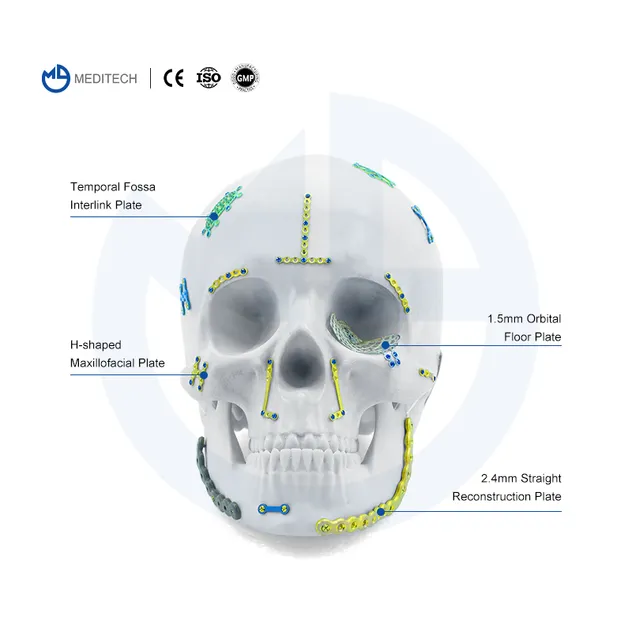
This Neurosurgery Restoration and Reconstruction System is a professional full-category cranial fixation implant system specially developed for neurosurgery craniotomy skull flap fixation, cranial defect reconstruction, traumatic skull fracture repair, cranial tumor postoperative reconstruction and maxillofacial cranial contour surgery. The system includes a full range of 0.6mm thickness titanium cranial fixation implants: interlink plates, temporal fossa plates, mastoid plates, minimally invasive titanium mesh, cranial pore plates, drainage cranial plates, and matching 1.5mm self-drilling titanium screws, fully meeting the full-process operational needs of various neurosurgery cranial fixation and reconstruction surgeries.
All implants are made of high-purity medical grade titanium with uniform 0.6mm thickness, which has excellent biocompatibility, corrosion resistance and mechanical stability, is fully compatible with MRI and CT examinations without artifacts, fully complying with international medical implant standards and clinical neurosurgery operation requirements.
| Product Name | REF No. | Thickness | Specification |
|---|---|---|---|
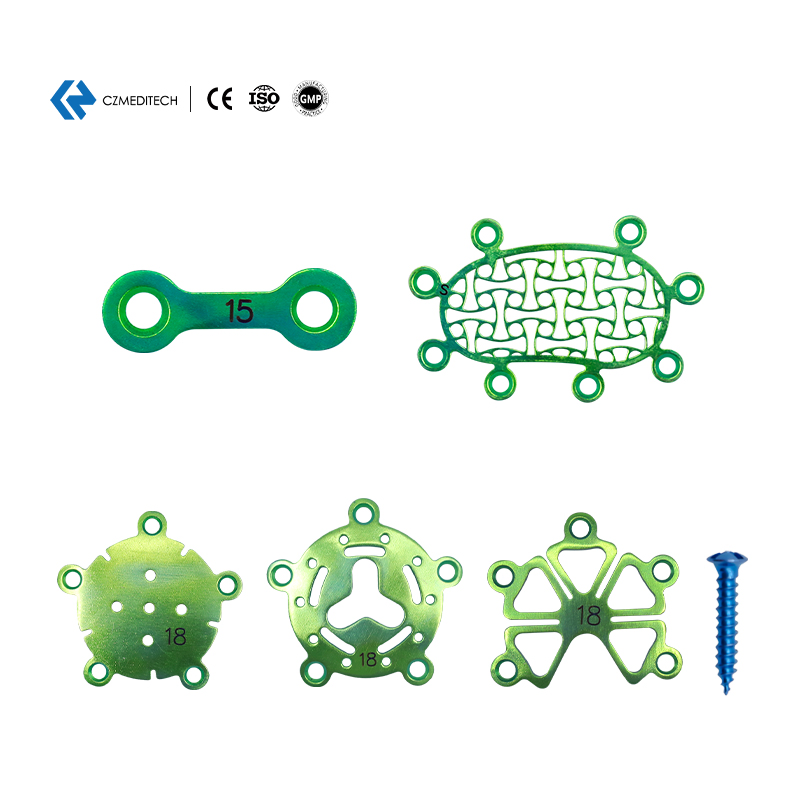
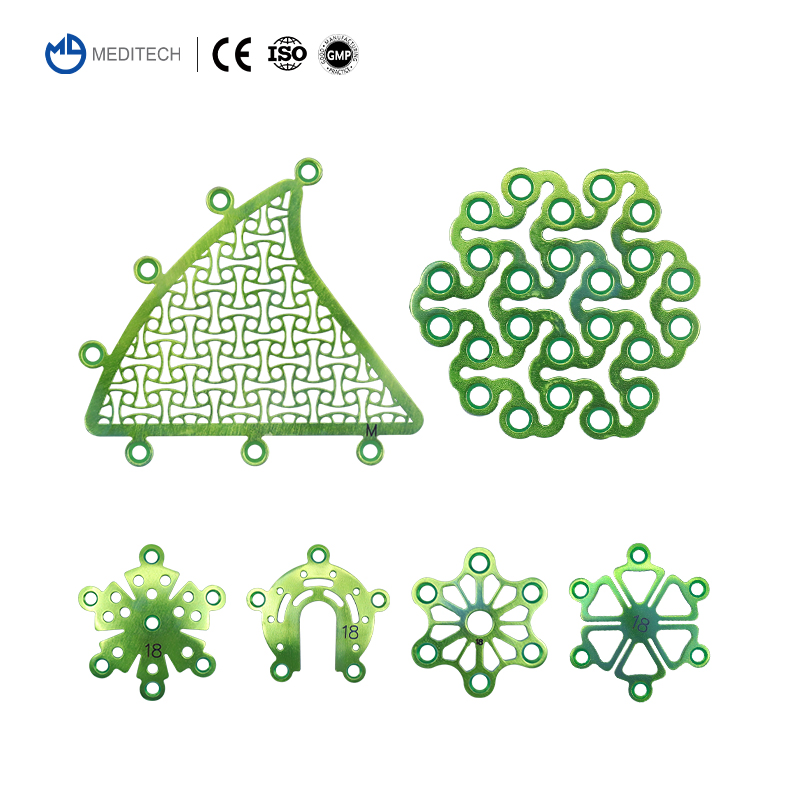
| Interlink Plate | 3000-0122 | 0.6mm | 2 holes 15mm |
| Temporal Fossa Interlink Plate | 3000-0125 | 0.6mm | M 30*30mm |
| Mastoid Interlink Plate | 3000-0128 | 0.6mm | Medium |
| Minimally Invasive Titanium Mesh | 3000-0130 | 0.6mm | 30*30mm |
| Cranial Interlink Plate-I | 3000-0131 | 0.6mm | 18mm |
| Cranial Pore Plate-III | 3000-0153 | 0.6mm | 16mm |
| Drainage Cranial Pore Plate-I | 3000-0157 | 0.6mm | Φ16*4mm |
| 1.5mm Self-drilling Screw | 2115-0141 | - | 1.5*4mm |