❓ Frequently Asked Questions
What material is used for the Minimally Invasive Spine System?
The system is manufactured from high-grade Titanium alloy, featuring a Micro-Arc Oxidation surface treatment for enhanced durability and biocompatibility.
What certifications do these orthopedic implants hold?
Our products are fully certified with CE and ISO13485 standards, ensuring they meet international quality and safety requirements for medical devices.
Is there a Minimum Order Quantity (MOQ)?
Yes, the MOQ is very flexible, starting at just 1 PC, allowing for both small-scale clinical use and larger procurement needs.
Do you offer OEM or ODM services?
We provide professional OEM and ODM services to customize the spine system according to specific surgical requirements or technical specifications.
What are the primary applications for this system?
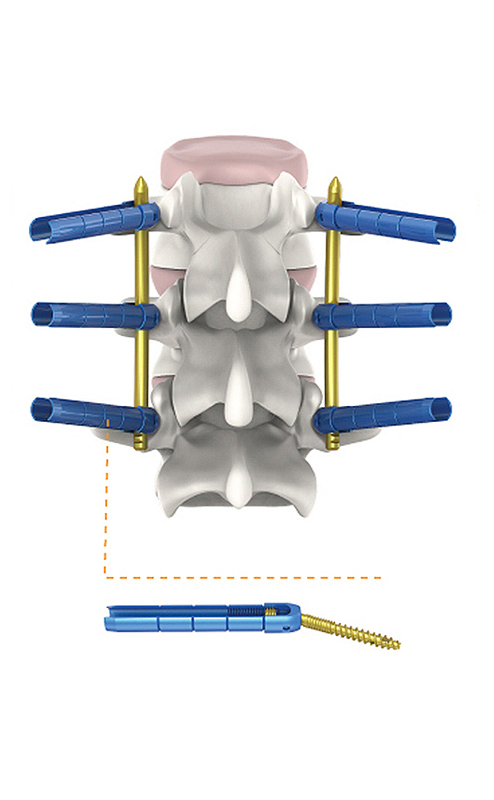
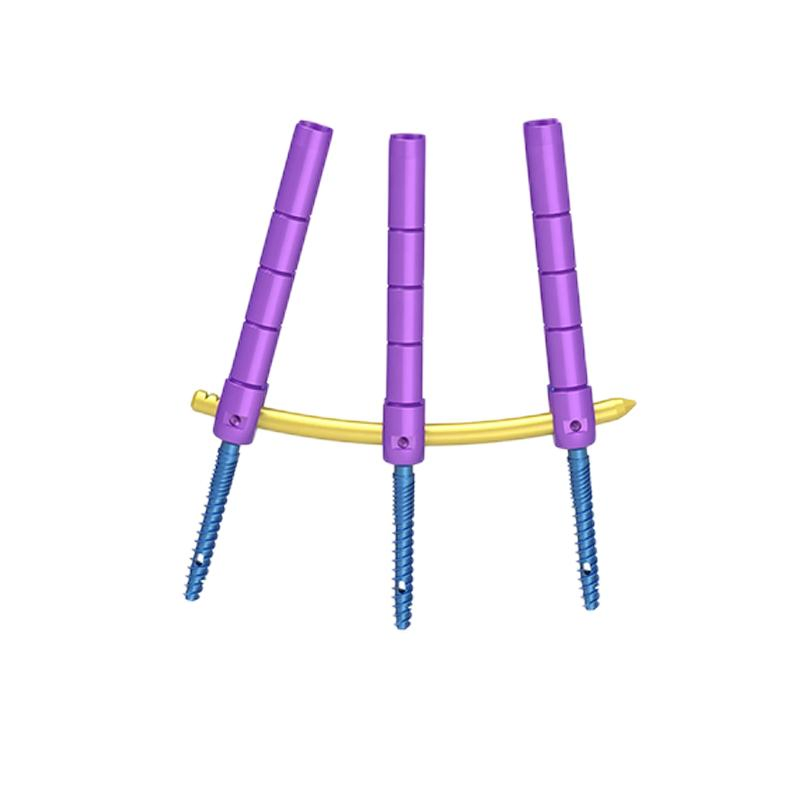
The system is specifically designed for bone fracture fixation in adults, particularly for minimally invasive spinal surgical procedures.
How is the product packaged for international shipping?
Each item is securely packed in PE bags, followed by an inner carton and a robust outer carton to ensure safety during air cargo transit via DHL, UPS, or FedEx.



 Medisage Medical
Medisage Medical


















