
1 / 5
 Medisage Medical
Medisage Medical










| Product Code | Product Name | Model | Standard Configuration Description |
|---|---|---|---|
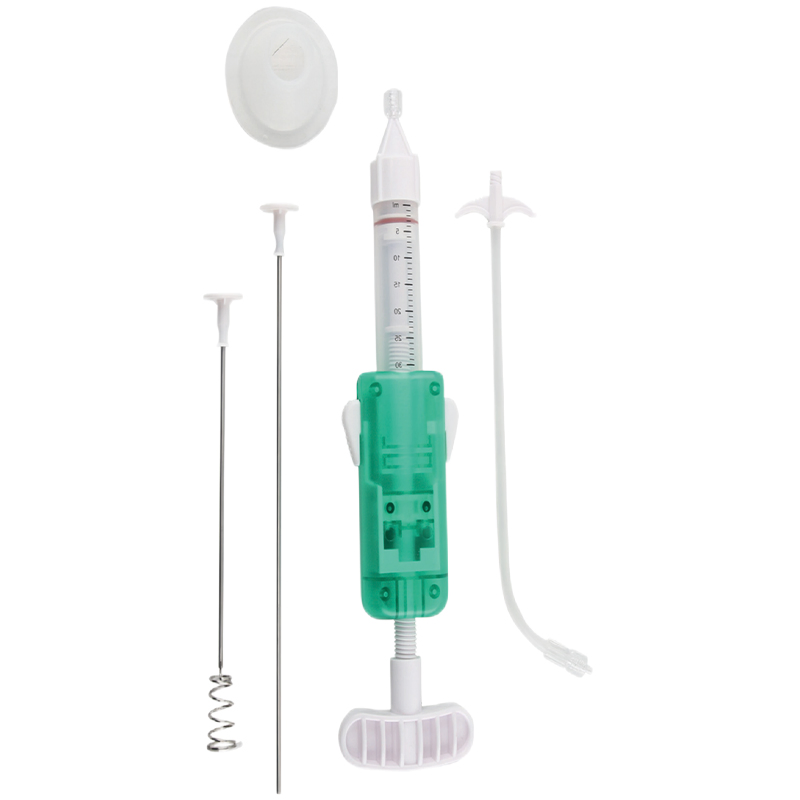
| 030020003 | Kyphoplasty Tool Kit | Type B (Lumbar Spine) | φ3.0 Percutaneous Access Device (1pc), φ3.5 Aiguille (1pc), Guide Wire (1pc), φ4.2 Expansion Cannula (1pc), φ3.5 Bone Cement Applier (4pc) |
| 030020002 | Kyphoplasty Tool Kit | Type D (Lumbar Spine) | φ3.0 Percutaneous Access Device (2pc), φ3.5 Aiguille (1pc), Guide Wire (2pc), φ4.2 Expansion Cannula (2pc), φ3.5 Bone Cement Applier (6pc) |
| 030020001 | Kyphoplasty Tool Kit | Type G (Lumbar Spine) | φ4.2 Percutaneous Access Device (1pc), φ3.5 Aiguille (1pc), Guide Wire (1pc), φ3.5 Bone Cement Applier (4pc) |
| 030030005 | Kyphoplasty Tool Kit | Type T1 (Thoracic Spine) | φ2.4 Percutaneous Access Device (2pc), φ3.0 Aiguille (1pc), Guide Wire (2pc), φ3.6 Expansion Cannula (2pc), φ3.0 Bone Cement Applier (6pc) |
| 030030006 | Kyphoplasty Tool Kit | Type T11 (Thoracic Spine) | φ2.4 Percutaneous Access Device (1pc), φ3.0 Aiguille (1pc), Guide Wire (1pc), φ3.6 Expansion Cannula (1pc), φ3.0 Bone Cement Applier (4pc) |
Integrated, one-step design for quick and efficient percutaneous access to bone and creation of bone tissue guide channels. This device helps to: