Frequently Asked Questions
What materials are used in the construction of this external fixator?
The system is manufactured using premium Titanium Alloy and Stainless Steel for the main structural components, along with medical-grade aluminum and carbon fiber to ensure excellent fracture visualization and strength.
Is the external fixator system reusable?
Yes, the system is designed to be reusable after proper sterilization and inspection, providing a cost-effective solution for medical facilities.
Which certifications does this medical device hold?
Our external fixator systems are fully certified with CE and ISO13485 standards, ensuring they meet international quality and safety requirements for orthopedic surgery.
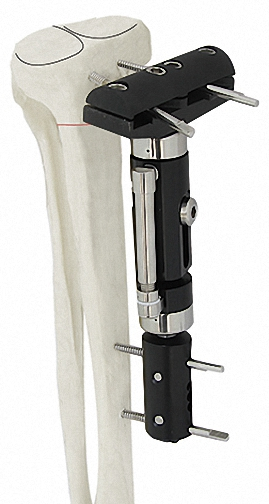
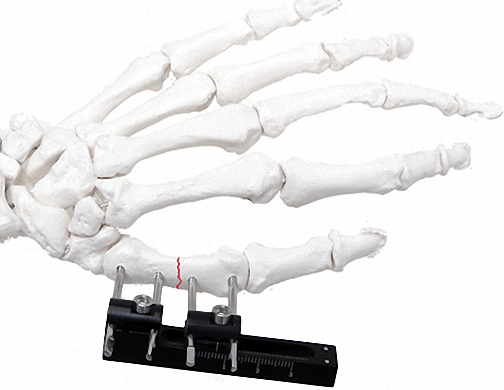
Can bone screws be arranged independently?
Yes, the modular design allows for the independent and precise arrangement of bone screws, which is essential for managing both proximal and distal fractures effectively.
What is the annual production capacity for these units?
We have a robust manufacturing capability with a production capacity of up to 500,000 pieces per year, ensuring steady supply for large-scale medical needs.
What types of joints can be treated with this modular system?
The system includes specialized modules for a variety of applications, including the Tibia Femur, Humeral & Tibial Lateral Epicondyle, Ankle Joint (Equinus), and Knee Joint.



 Medisage Medical
Medisage Medical

















